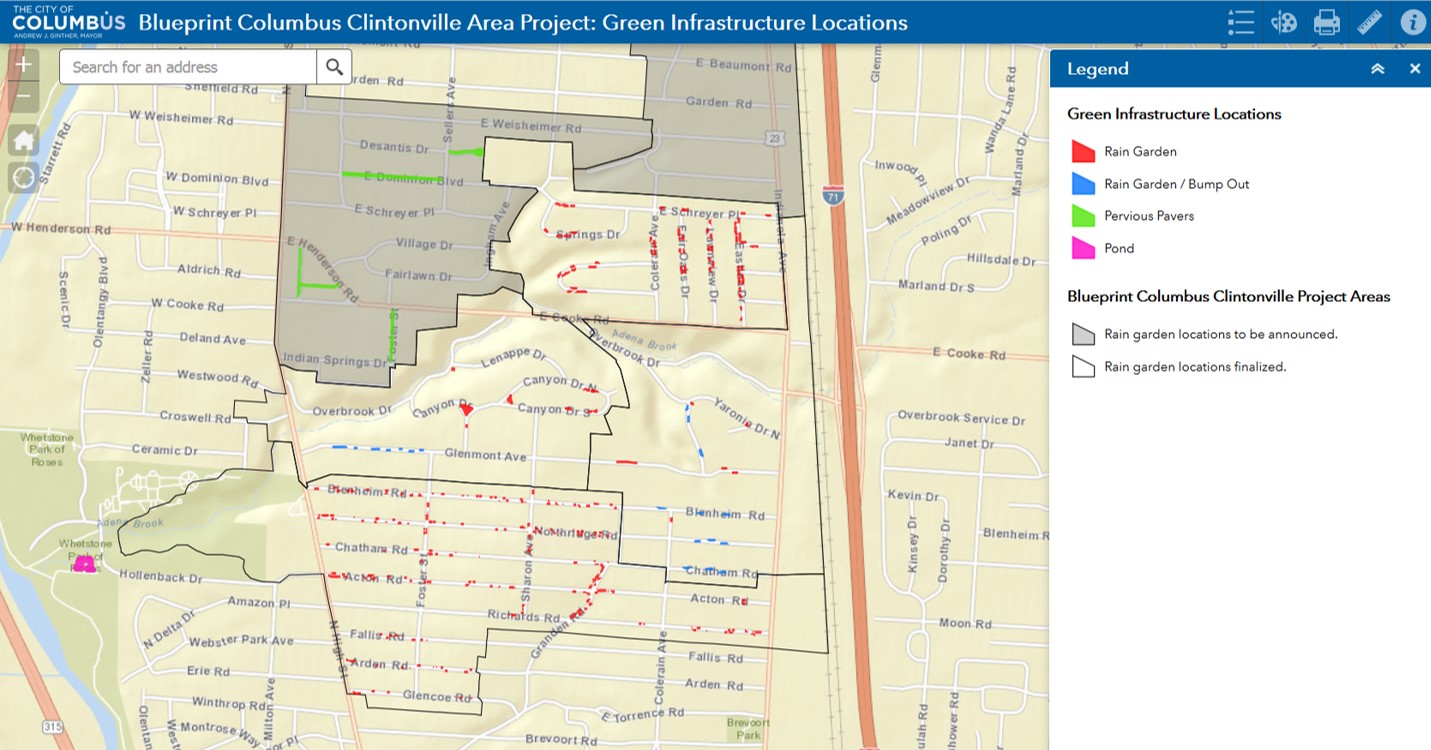
Brandon Sinn, PhD graduate from the OSU herbarium, now a postdoctoral fellow at West Virginia University, recently published a paper on molecular work he did to better understand the evolution of genomes in Asarum (Aristolochiaceae), commonly known as wild ginger. The work was done in collaboration with Dylan Sedmak, an OSU undergraduate student, Lawrence Kelly, Associate VP of Science, New York Botanical Garden and John Freudenstein, Professor and Chair of EEOB and Brandon’s PhD advisor.
We interviewed Brandon to get a better understanding of his research findings:
Brandon: “Evolutionary relationships in the flowering plant genus Asarum served as the focus of my dissertation research, and I continue to study the group. In this particular project we studied six Asarum species, which each represent one of the six major evolutionary lineages within the genus.
Asarum is a poorly-understood genus of approximately 115 species found in temperate forests across Asia and North America. Some Asarum species are common and widespread across the continents where they are found, while the majority have highly restricted ranges – for example, one species is known only from a single gorge in North Carolina and others are found in only a few counties in the southeastern United States.
During the course of sequencing DNA for my dissertation research, I realized that the genes of some Asarum species were not in the expected order. This departure from expectation was surprising, since the clade, or evolutionary neighborhood, that Asarum belongs to is very old and had been partially characterized as having slowly-evolving and highly conserved genomes. For example, the genome of another member of the same clade has been called a “fossil” genome. It was because of this unexpected observation that we decided to sequence complete genomes from one species from each of Asarum lineage. ”
This lead to the following research questions: Note: A plastome is the genome of a plastid, the organelle responsible for photosynthesis in plants.
1) Have the plastomes of all Asarum species been destabilized and their gene order rearranged?
2) Is the plastome of Saruma henryi (commonly called upright wild ginger), the closest relative of Asarum, of typical arrangement or is it more like that of Asarum?
3) Can we understand how the ordinarily highly conserved and stable plastomes become destabilized by comparing the plastomes of many Asarum species to that of Saruma henryi?
What should we know to understand this research?
Brandon: “Each plant cell contains at least one copy of three distinct genomes. It is easy to imagine that each cell has a copy of the plant’s genome, but many people forget that two types of plant organelles, mitochondria and plastids, also have their own genomes. Plastids, from which chloroplasts develop, have a very small genome that is relatively easy to completely sequence and the sequence of more than 2,000 are publicly available today. The sequencing of thousands of plastomes has resulted in several general trends: 1) plastomes change more slowly than the plant’s own genome; 2) the plastome is made up of three functional regions, the small single copy, large single copy, and inverted repeat regions; 3) the physical order of genes is highly conserved across even distantly related species; 4) there is very strong selective pressure on the preservation of photosynthesis, which most likely constrains the evolution of plastomes in green plants. Our knowledge of the typical layout of the plastid genome, or plastome, has long been relied upon to sequence DNA in order to study plant evolutionary relationships. Traditional DNA sequencing techniques require prior knowledge of the order of genes or regions of a genome. If this order is not as predicted, then the DNA sequencing will fail.”
What method did you use to study your research question?
Brandon: “For this study, we sequenced entire plastomes from six Asarum species and that of Saruma henryi, the closest relative of Asarum. Since traditional DNA sequencing is not useful in destabilized and dynamically rearranged genomes, and we wanted to sequence entire plastomes that we hypothesized were rearranged, we needed to use a technology called massively parallel sequencing. A major advantage of massively parallel sequencing is that a researcher can extract DNA from a tissue, break the DNA into short pieces, and simultaneously sequence all of these fragments without prior knowledge of their physical relationship to one another. The resulting millions of DNA sequences are then assembled, much like a puzzle, using specialized software. The assembled plastomes can then be compared.”
Brandon explains one of the key figures in his manuscript:

A cruciform DNA structure that has likely destabilized a region of the plastome in Asarum species. The end of the ndhF gene is shown in red. Structure courtesy of Eric Knox.
DNA is made of only four chemicals (which we abbreviate as the letters A, T, C and G) and is not entirely unlike a spiral staircase, where each handrail is a string of these letters. Holding this structure together are bonds that form between certain letters – A-T and G-C. We call these letters nucleotides. Sometimes the nucleotides making up DNA cause the molecule to form complex shapes, such as the cruciform structure shown here. Cruciform, or cross shaped, DNA structures form when the same nucleotides are repeated very close to one another, which is depicted in the vertical “stems”.
Cruciform DNA structures can be difficult for the molecular machinery in cells to work with. For example, sometimes molecules that interact with DNA get stuck on the stems, and these structures compromise the integrity of the DNA molecule. When these structures break, which you can imagine by separating the red and black halves of DNA for Saruma henryi, the cell tries to put them back together. But, repairing DNA does not always work perfectly. The results of our research suggest that faulty repairs made to this DNA structure throughout the plastomes of Asarum species have resulted in varying degrees of DNA duplication. Notice that the ndhF gene (shown in red) is typically at one end of the small single copy region, as shown on the Saruma henryi plastome. In Asarum, this gene often has a long stretch of nucleotides that can be “pasted before or after it. In other Asarum plastomes, such as Asarum canadense, we find that all of the small single copy region has been duplicated. The duplication of the formerly single copy region is most likely due to faulty repair of the cruciform DNA structure, where identical strings of nucleotides close to one another led to bonding of two identical DNA regions (as seen in the Asarum canadense cruciform structure).”
Why is this research important?
Brandon: “When you learn about DNA in high school science classes, everything sounds very concrete and well understood, but even gene function in humans is not exhaustively understood. Our basic knowledge about how genes and genomes evolve is in a constant state of improvement. This knowledge is necessary for future breakthroughs in genome engineering, evolutionary and conservation biology, and improving genome stability. Just as it is important to understand biodiversity at the level of species, it is equally important to understand genomic diversity – the content and structure of genomes, in order to understand how mutations in particular regions of genomes can lead to genome-scale changes over deep time and how these changes affect evolutionary lineages.”
What should you take away from these findings?
1) Just because a species is a member of a very old evolutionary lineage, we should not expect that it is a living fossil and that its genome has changed little.
2) A plastome can function even when gene order is changed and more than half of its genes are present more than once.
3) Small, likely randomly generated repetitive motifs in DNA sequence that is not part of a gene can decrease genome stability, and lead to genome rearrangement and gene duplication.
*******************************************************
Wow, we are now certainly asking questions and getting answers with new techniques that we could not have imagined decades ago. If you want to follow Brandon’s further research, click here.
About the Authors:  Brandon Sinn earned his Ph.D. in 2015 from the Department of Evolution, Ecology and Organismal Biology, where he was a member of the Freudenstein Lab in the Museum of Biological Diversity. Brandon has held a postdoctoral research position at the Pfizer Plant Research Laboratory of the New York Botanical Garden, where he worked on the Planteome Project. He is currently a postdoctoral fellow in the Department of Biology of West Virginia University where he studies orchid genome evolution as a member of the Barrett Lab.
Brandon Sinn earned his Ph.D. in 2015 from the Department of Evolution, Ecology and Organismal Biology, where he was a member of the Freudenstein Lab in the Museum of Biological Diversity. Brandon has held a postdoctoral research position at the Pfizer Plant Research Laboratory of the New York Botanical Garden, where he worked on the Planteome Project. He is currently a postdoctoral fellow in the Department of Biology of West Virginia University where he studies orchid genome evolution as a member of the Barrett Lab.
 Angelika Nelson is the curator of the Borror Laboratory of Bioacoustics and the social media manager for the museum.
Angelika Nelson is the curator of the Borror Laboratory of Bioacoustics and the social media manager for the museum.
Follow us also on Facebook.
Reference:
Sinn, B. T., Sedmak, D. D., Kelly, L. M., & Freudenstein, J. V. (2018). Total duplication of the small single copy region in the angiosperm plastome: Rearrangement and inverted repeat instability in Asarum. American journal of botany, 105(1), 71-84.