Echinoderm Environment:
Echinoderms are the largest phylum with no freshwater or terrestrial forms. Echinoderm environments must be marine, as in saltwater, for the echinoderm to survive. Within marine environments, the conditions echinoderms live in can vary greatly. Environments range in water temperature, water depth, water movement and the different organisms surrounding the echinoderms. Water temperatures can range from arctic temperatures to tropical temperatures. Water depth and movement somewhat tie in together in the idea that the more shallow the water, the more likely the water will be moving. Deeper water is sometimes less affected by rain and storms and therefore does not affect the echinoderms as much. Water movement could impact the echinoderms by moving them or destroying their habitats. Depending on the species the water depth and movement will vary. Echinoderms are generally found in shallow water near shores or in reef environments but can also live in great depths of water. Almost all echinoderms are benthic, meaning they live on the seafloor, but some sea lilies can swim for some distance while some sea cucumbers float throughout the water (Xiaofeng, Hagdorn, & Chuanshang, 2006). Below is a photo of a tank for marine animals. This tank contains brittle stars, sea cucumbers, and sea urchins along with non-echinoderm animals. This marine tank is kept in conditions to reflect the echinoderm’s natural habitat. The tank is salt water and is kept at 78°F. In the tank are rocks, plants and other things to help give shelter and protection to the animals.
This picture is of a tank in a classroom at Ohio State University. In this tank there is a species of brittle stars, sea urchins and sea cucumbers. There are also other animals such as a shrimp and some small fish. Not to mention the plants and sponges present.
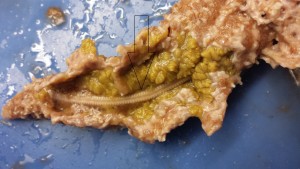
Brittle stars (Ophio coma) lead a cryptic lifestyle. They avoid predation by spending much of their day hiding within holes in coral and rocks found in their environment (Zubi, 2015). In the picture shown below, a brittle star is almost completely concealed under a sponge. Likewise, sea urchins’ main defense from predators is creating a burrow in the ground to leave less of their body exposed to predation. When sea urchins are found in environments not conducive to burrowing, like the marine tank shown above, they have been know to cover themselves with objects found in the environment for protection (Carefoot, 2010). The sea urchin shown below is exhibiting this trait, covering itself with shells from other organisms.


The photo on the left is of a brittle star hiding out under a sponge in the tank at Ohio State. The photo on the right is of a sea urchin in the same tank carrying around a shell of another organism.
Lifecycle:
Though many Echinoderm species undergo sexual reproduction, a few species reproduce asexually. The bulk of echinoderm species are diecious, meaning that there are male and female individuals. The type of reproduction that an echinoderm goes through depends on the species and the environment (Mulcrone, 2005).
Sexual Reproduction:
Sexual reproduction refers to the combination of genetic material from two separate individuals, of different sexes, resulting in a new organism. The males provide the sperm and the females provide the eggs for reproduction. The sperm and eggs are propagated into the water column, and fertilization generally happens in the open water. Certain species of sea stars, brittle stars, and sea cucumbers go through internal fertilization. This is a recent observation and little information is known about this phenomenon. The release of the eggs and sperm is synchronized in two different ways. Some species have a breeding season where individuals congregate to increase the chances of fertilization. Some species’ breeding season may be affected by the lunar cycles. Commonly, echinoderms will leave their eggs and provide no parental care to the eggs (Young & Eckelbarger, 1994). Some species such as the Slender Sea Star (Leptasterias tenera) brood their eggs. This means that they carry their eggs with them until they are ready to hatch. This species of starfish carries the eggs in their stomach but other echinoderms carry the eggs in places such as in sacks under their arms, their coelom (main body cavity containing the organs) or in special egg cavities (Hendler & Franz, 1982).
Development:
When sexual reproduction occurs, the resulting eggs go through either direct or indirect development. When indirect development occurs, the fertilized eggs of echinoderms will develop into larvae known as planktonic larvae. In most cases, this stage occurs when the fertilized egg consists of a lower yolk volume. The resulting larvae ends up assimilating into the surrounding plankton community. They will start feeding on numerous smaller organisms in an effort to meet the energy requirements needed to perform metamorphosis, and achieve their juvenile stage of their adult form. After a period ranging from one to three weeks, these bilateral symmetric larvae begin their metamorphosis, during which they will develop five water vascular canals that will eventually give them their characteristic pentaradial symmetry.
In contrast, during direct development, fertilized eggs will contain a larger yolk volume. Due to this, they are able to be sustain their life cycle development solely on their yolk, saving them from expending energy feeding, and also allowing them to bypass the larval stage of development. In this developmental strategy, often times the female parent will be involved in rearing the juveniles. In these instances, the female will hold the young juveniles near its mouth, on the underside of its body, or even in specially developed pouches that are found on the upper side of the body. This allows the female to provide predation protection to its young until they fully developed. (Pawson, 2014)
Asexual Reproduction:
Echinoderms are capable of performing asexual reproduction, and the most common way they achieve this is through division of body parts or body sections, which is called fragmentation. What happens in this reproductive strategy is that an echinoderm may deliberately or accidently detach a limb or body section. This may happen due to possible predation, an unforeseen accident, or in some cases with species of starfish, it separates itself by pulling opposing limbs in opposite directions until the individual splits. (Pawson/Encyclopedia Britannica, 2014)
The reason this works without permanently harming the individual is because of a unique process in which they are able to seal areas of exposed tissue where a section has been detached, and then immediately it begins regenerating new tissue to replace that section. Not all classes accomplish this strategy in the same manner, as multiple solutions seems to have arisen within each class. In species of starfish and brittle stars, this process requires that part of the central disk be included with each separate segment in order to regenerate into a complete individual. In species of sea cucumber, not only do they seal off the “wound” from where they split in half, the resulting two regenerating parts have to go through a tissue reorganization in order to successfully complete the process. (Speer and Waggoner 1999) In sea lilly and other starfish species, they can simply detach a limb or body section regardless of what part is present and either regenerate the missing limb, or in the limbs case, regenerate an entire new body. While impressive, this asexual reproductive method has shown itself to be pesky for habitat management teams, as complete removal of sea stars and sea lilies can prove to be very difficult. (Pawson/Encyclopedia Britannica, 2014)
Echinoderms throughout evolutionary time:
The first true echinoderms appear in the fossil record in the Cambrian period, which began about 560 million years ago. During the pre-Cambrian period, echinoderm ancestors are not believed to have had radial symmetry (Zamora, 2012). Echinoderm ancestors in the pre-Cambrian are also thought to have been soft-bodied with unmineralized plates, which contrasts with all true echinoderms, starting in the Cambrian, who have hard mineralized skeletons which provide structure and protection (Speer, 1999). The earliest echinoderms are thought to have been deposit feeders, which intake and extract nutrients from the sediment on the floor of the ocean. This varies from the current method of feeding found in echinoderms today, suspension feeding, in which echinoderms eat particles found suspending in their environment (Steele). The first echinoderms in the Cambrian period had both radiate and asymmetric forms and a similar water vascular system to those found today, including one coelom (Zamora, 2012).
During the mid-Cambrian, the species richness, or the number of species, increased by fifteen percent. In the Ordovician period, starting around 510 million years ago, the earliest echinozoans, starfish, and brittle stars appear (Baumiller, 2015). After the Great Ordovician Biodiversification Event (GOBE), suspension-feeding organisms, like the echinoderms dominated ocean floor habitats. Three echinoderm classes from the Cambrian went extinct during this period, six classes survived from the Cambrian, and eleven new classes appeared after the GOBE. No new classes of echinoderms have emerged since the GOBE (Baumiller, 2015).
Since the Ordovian period, the number of classes has steadily declined. Though the classes of sea lilies and blastoids dominated the echinoderm phylum through most of the Paleozoic period, by the end of the Permian period, which ended 250 million years ago, all of the blastoids and almost all of the sea lilies were extinct. From the Mesozoic period, which directly followed the Permian period, to today, there have been only five classes of echinoderms, the Sea Lilies (Crinoidea), Starfish (Asteroidea), Brittle stars (Ophiuroidea), Sea Urchin (Echinoidea), and Sea Cucumbers (Holothuroidea) (Baumiller, 2015).
Diversity:
With 7,000 living species, echinoderms are very diverse (Mulcrone, 2005). Echinoderms vary in physiology and morphology such as shape, size (width and length), color and locomotion. Amongst the five main classes of echinoderms, probably the most obvious difference is the shape of the body. For example sea stars have a star looking shape while sea urchins are spherical, sea biscuits and sand dollars are round, and sea cucumbers are longer and more caterpillar-like. Below is a photo, from left to right, of a brittle star skeleton, sea cucumber and sea urchins remains, followed by live specimens of sea cucumbers, sea urchins, sea biscuits, and sand dollars. The diversity in the body shape can clearly be seen.
In the photo above you can not only see the diversity in body shape but also the color and size. The brittle star in the photo is an Orphiarachnella ramsayi and was found on a reef in Sydney Australia. These brittle stars can range in colors from black and white to having some pink, red and green as well (O’Hara, 2015). Compared to the sea cucumber that is bright orange, the color obviously varies. The size between these three specimens can range from 1-10 inches. Diversity in echinoderms is not only between the five classes but between the species in each class. For example sea stars vary in size, color and number of appendages. Below is a photo of three very different sea stars.

This photo shows the variance in size, color, and number of limbs that can be on just sea stars alone. The starfish on the left was about seven or eight inches across while the starfish on the right was less than an inch. The color variation is also shown.
Shown above is a blue sea star, Linckia laevigata.
As seen in the photo the sea star on the left is larger and has only five appendages while the sea star on the right is much smaller and has six appendages. The sunflower starfish (Pycnopodia helianthoides) can have between 16 and 24 arms and can be up to a meter in diameter (Sustainability Species Identification). This just shows the diversity of appendages in sea stars. The sea cucumber and sand dollar have no appendages at all. One other notable difference that has occurred between echinoderms is their locomotion, or how they move. For example sea stars move by using their tube feet to attach to the ground and sort of walk. Brittle stars on the other hand use their appendages to push themselves from place to place. Finally, sea cucumbers can pull themselves along somewhat like a snail and even some species can swim (Mah, 2012). Sea urchins, primarily move using their tube feet, however, they can also use their spines for fast movement, as shown in the video below. The next two videos show a brittle star, a chocolate chip sea star, and a sea urchin moving.
This video shows how a brittle star uses its limbs, as opposed to tube feet, to move around the tank.
This is a timelapse of a starfish moving around on top of an oyster.
https://www.youtube.com/watch?v=s7qagaFoHyk&feature=youtu.be
This video shows a long-spined sea urchin, Diadema setosum, moving using its spines. Sea urchins primary mode of locomotion is their tube feet, but they occasionally use their spines for movement.
This is just a short overview of the diversity of echinoderms that has developed over millions of years.
Human impact:
Human beings as we continue to grow in numbers have a lasting impact on many different animal species, however, it is becoming more evident of our impact on the oceans and the animal species that call the oceans home. This section will specifically cover the effects humans have on echinoderms but many of these effects have a cascading effect on many different animals.
The fishing industry has a large impact on the survival of echinoderm species. The fishing industry removes substantial amounts of the echinoderms’ diet from the oceans including, clams, mussels, and oysters. This reduces the food available to the echinoderms. The pet trade also has several direct effects on many species of echinoderms. A big part of the pet trade industry involves collecting corals and live rock for sale in many countries all over the world. The coral and live rock make up the habitats of numerous echinoderms and removing them effectively leaves the echinoderms unable to protect themselves. Also, humans pull all echinoderms (starfish, sea urchins, sea cucumbers, sea lilies and brittle stars) out of their natural habitat to be directly sold in the pet trade. Both of these problems affect the number of echinoderms in the wild. Taking rock from reefs removes a lot of habitat that these animals rely on, and removing them from areas completely diminishes the number of breeding animals.
Humans play a large role in the habitat destruction affecting many echinoderm species. Human have a large carbon footprint, and as we are releasing large amounts of carbon dioxide into the atmosphere, the pH of the oceans is slowly rising. This slight increase in pH is not detrimental to adults in all species currently, but it has a significant impact on the more sensitive species of echinoderms. Also, these environmental changes do affect all echinoderms during their developmental stage when they are the most susceptible to environmental changes. This effect can be severe; a change of just 0.2 units of pH leads to a 100% mortality rate in 8 day old common brittle stars (Ophiothrix fragilis) (Dupoint, 2009).
Conclusion:
Echinoderms are found all over the world and play a major role in the animal kingdom and the environment. They help to keep algae growth down, feed other animals, and feed people in certain countries. People need to understand the importance of echinoderms. If too many are captured or we keep destroying their habitats, it could affect more than just the echinoderm’s future. As a keystone species in many ecosystems, the loss or elimination of species of Echinoderms could drastically and permanently affect numerous marine habitats around the world, as well . They are a diverse family, with 7,000 currently known species of echinoderms ranging from sea stars to sea cucumbers, and with the scientific community making new discoveries using ever developing technology, we could see many more species discovered in our lifetime.
Work Cited:
Baumiller, T. (2015). Fossil record of Echinoderms. Retrieved from https://scripps.ucsd.edu/centers/echinotol/about-echinoderms/fossil-record-of-echinoderms/
Carefoot, T. (2010). Learn About Sea Urchins: Predator and Defenses. Retrieved from http://www.asnailsodyssey.com/LEARNABOUT/URCHIN/urchDefe.php
Dupoint, S., & Thorndyke, M. (2009). Impact of CO2 -driven ocean acidification on invertebrates early life-history – What we know, what we need to know and what we can do.Biogeosciences Discuss, 6, 3109-3131. accessed 29 March, 2015.
Echinodermata. (n.d.). Retrieved from http://www.bio200.buffalo.edu/labs/echinoderms.html
Echinoderms. (n.d.). Retrieved from http://www.oceaninn.com/wildlife/echinoderms.htm
Hendler, G and Franz, D. (June, 1982). Washington, DC: Smithsonian Institution and New York, NY: Brooklyn College.
Mah, C. (2012, Sept. 18). Deep-Sea Swimming Sea Cucumbers and the “most bizarre holothurian species in existence”! Retrieved from http://echinoblog.blogspot.com/2012/09/deep-sea-swimming-sea-cucumbers-and.html
Mulcrone, R. (2005). Echinodermata (sea stars, sea urchins, sea cucumbers, and relatives). Retrieved from http://animaldiversity.org/accounts/Echinodermata/
Pawson, D. (2014, July 24). Echinoderm. Retrieved from http://www.britannica.com/EBchecked/topic/177910/echinoderm
O’Hara, T. (2011). Brittle Star. Retrieved from http://portphillipmarinelife.net.au/species/6607
Speer, B., & Waggoner, B. (1999, September 15). Echinodermata: Fossil Record. Retrieved from http://www.ucmp.berkeley.edu/echinodermata/echinofr.html
Speer, B., & Waggoner, B. (1999, September 15). Echinodermata: Life History and Ecology. Retrieved from http://www.ucmp.berkeley.edu/echinodermata/echinolh.html
Steele, M. (n.d.). Ecology of Soft-Sediments. Retrieved from http://www.csun.edu/~msteele/classes/marine_ecology/lectures/15_soft-sediment ecology.pdf
Sustainability Species Identification. (n.d.) Sunflower sea star. Retrieved from http://www.nmfs.noaa.gov/speciesid/fish_page/fish6a.html
Xiaofeng, W., Hagdorn, H. and Chuanshang, W. (2006). Pseudoplanktonic lifestyle of the Triassic crinoid Traumatocrinus from Southwest China. Retrieved from http://onlinelibrary.wiley.com/doi/10.1080/00241160600715321/abstract;jsessionid=9342400ED743C5DFC4E653363D1CDAF0.f02t02
Young, Craig M.; Eckelbarger, Kevin J. (1994). Reproduction, Larval Biology, and Recruitment of the Deep-Sea Benthos. Columbia University Press.
Zamora, S., Rahman, I., & Smith, A. (2012). Plated Cambrian Bilaterians Reveal the Earliest Stages of Echinoderm Evolution. PLoS ONE, 7(6).
Zubi, T. (2015). Invertebrates: Echinoderms. Retrieved from http://www.starfish.ch/reef/echinoderms.html