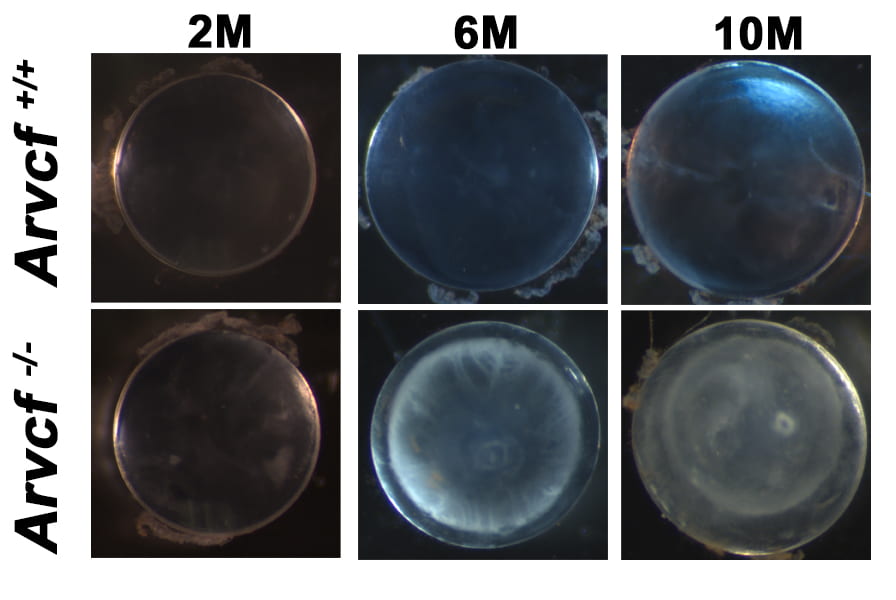
Identifying the etiology of lens disease
The function of the lens requires the maintenance of its transparency and refractive properties throughout one’s lifespan but those mechanisms responsible are not well understood. With age, the lens can develop opacities, or cataracts, which disrupts vision and requires surgery and implantation of an artificial lens. We have discovered that a specific type of age-dependent cataracts (cortical cataracts) can occur with the genetic disruption of a critical protein found within the adherens junctional complex of mice. This gene/protein is known as ARVCF and it is strongly expressed in lens fiber cells and is localized to the junctions, particularly those located within the interlocking protrusions. These structures interlock with neighboring cells and are thought to maintain the organization and mechanical properties of the lens. Importantly, these structures are preferentially located in the region of the lens that develop opacities during age-dependent cortical cataracts. This current project is focused on identifying what physiological functions within lens fiber cells are dependent on Arvcf and which of those fail during the progression of cortical cataract formation.
Mechanisms of ocular morphogenesis
Generation of the eye, more so than many organs, requires precise control of its shape for optimal function. Obtaining knowledge of how the eye and lens is constructed during embryonic development is therefore important to help describe the nature of ocular abnormalities that lead to major structural defects or more subtle changes that alter vision.
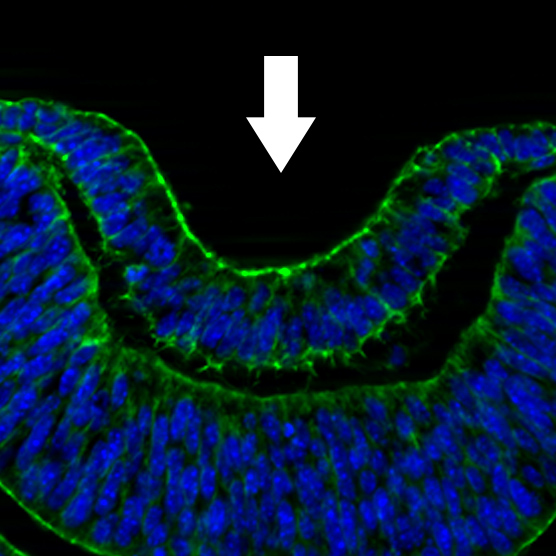
Lens invagination
An example of a morphogenetic event required for the generation of organs is epithelial invagination. This process drives the inward bending of epithelia of several early organ systems including that of the lens placode during early ocular development. We have observed that a major driving force of lens placode invagination is the contraction of supracellular actomyosin rings that encircle the border of the lens placode. The mechanisms that drive the formation of these supracellular structures, their alignment with neighbors, and the molecules that trigger contraction are not understood in the context of any vertebrate epithelia. We are currently undergoing experimentation to test hypotheses regarding the identity of the responsible cytoskeletal/junctional proteins for these processes.
Shroom3-dependent ocular development
Another type of birth defect that affects the normal shape of the eye is called a coloboma, a condition where the early retinal structures (the optic cup) fails to normally shape itself at its ventral portion which can lead to defects in iris and retinal development. This disorder ranges in severity from being relatively benign to significant visual impairment. One of the cytoskeletal proteins we study is called Shroom3 and mice lacking the encoding gene develop a coloboma, making these mice a fantastic model to elucidate the mechanisms that lead to this birth defect. Shroom3 plays a role in several ocular structures including the lens, retina, lacrimal gland, and the cornea and current projects are focused on determining the role of Shroom3-dependent processes play in shaping their development.
Lens fiber cell morphogenesis
Lens fiber cells are unique epithelial cells that are extremely long, hexagonally arranged, and are decorated with numerous protrusive structures. These characteristics are thought to promote transparency, adhesiveness, and tissue homeostasis, however, the basic mechanisms of generating the morphology of lens fiber cells are not well understood. Conditional and conventional mouse knock-outs are currently being utilized to elucidate the role of specific genes that regulate lens fiber cell morphology. High-resolution immunofluorescent microscopy is being utilized to uncover hidden details of these processes.