The INK-ARF Pathway
The Poi Lab has focused on the cancer biology and preclinical development of anticancer agents for the treatment of head and neck cancer. Our research incorporates clinical pharmacology principles (pharmacokinetics, pharmacodynamics, drug metabolism, drug transport, drug-drug interactions) in drug development. Our lab has been studying the genetics and roles of the CDKN2A/2B genes and related pathways in human cancers. The CDKN2A/2B locus is fascinating because it codes for 3 tumor suppressors, namely P14ARF, P15 and P16.

RD: the regulatory sequence of the INK4-ARF locus; e1, e2: exons 1 and 2 of p15; in1, intron 1 of p15; E1β, E1α, E2, E3: exons 1β, 1α, 2, and 3 of p16; IN1: intron 1 of p16. The orange line and boxes represent the ANRIL gene (antisense non-coding RNA in the INK4-ARF locus) and its exons. Of note, ANRIL is transcribed from an unidentified promoter element close to E1β in an antisense direction with respect to p15, p14ARF, and p16 transcripts. The cross bar indicates negative regulation. PRC1/2, Polycomb repressing complexes 1 and 2. Sizes of coding regions and proteins are not in proportion strictly. The question mark (?) indicates that the functioning pathways of P12 and P10 remain to be defined.
(Reference: Poi MJ, Knobloch TJ, Li J. Deletion of RD(INK4/ARF) enhancer: A novel mutation
to “inactivate” the INK4-ARF locus. DNA Repair (Amst). 2017 Sep;57:50-55)
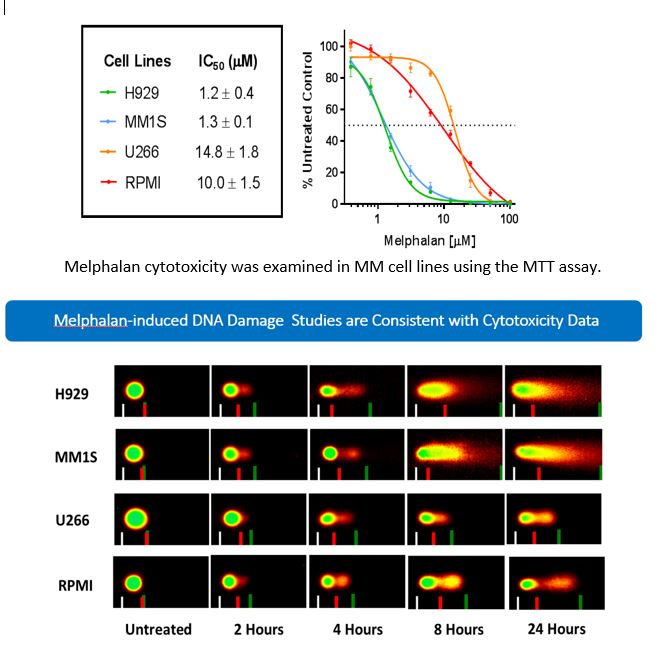
DNA Repair Pathways in Multiple Myeloma
Using similar approach, that is integrating clinical pharmacology principles (pharmacokinetics, pharmacodynamics, drug metabolism, drug transport, drug-drug interactions), our group is also interested in understanding the roles of the DNA repair pathway genes in multiple myeloma patients and treatment response to high dose melphalan (an alkylating agent) in the setting of autologous stem cell transplant. High-dose melphalan (HDM) followed by autologous hematopoietic stem cell transplant (auto-HSCT) is currently a cornerstone treatment for multiple myeloma (MM). However, interpatient variability in melphalan pharmacokinetics (PK) and pharmacodynamics (PD) contribute to both excessive adverse effects and inadequate dosing resulting in treatment failure. The primary factors responsible for melphalan interpatient variability are largely unknown but numerous studies have suggested that variation in inter-individual DNA repair capacity may partially explain the variability. Our long-term goal is to elucidate, validate, and incorporate factors (including genetic factors) responsible for melphalan interpatient variability into a PK/PD model that physicians can use to personalize melphalan dose.