
After a long week, the Fredrick lab enjoyed a nice happy hour at Budd Dairy Food Hall in downtown Columbus.

After a long week, the Fredrick lab enjoyed a nice happy hour at Budd Dairy Food Hall in downtown Columbus.
 Abstract
Abstract
Ribosome biogenesis occurs co-transcriptionally and entails rRNA folding, r protein binding, rRNA processing, and rRNA modification. In most bacteria, the 16S, 23S, and 5S rRNAs are co-transcribed, often with one or more tRNAs. Transcription involves a modified RNA polymerase, called the antitermination complex, which forms in response to cis-acting elements (boxB, boxA, and boxC) in the nascent pre-rRNA. Sequences flanking the rRNAs are complementary and form long helices known as leader-trailer helices. Here, we employed an orthogonal translation system to interrogate the functional roles of these RNA elements in 30S subunit biogenesis in Escherichia coli. Mutations that disrupt the leader-trailer helix caused complete loss of translation activity, indicating that this helix is absolutely essential for active subunit formation in the cell. Mutations of boxA also reduced translation activity, but by only 2- to 3-fold, suggesting a smaller role for the antitermination complex. Similarly modest drops in activity were seen upon deletion of either or both of two leader helices, termed here hA and hB. Interestingly, subunits formed in the absence of these leader features exhibited defects in translational fidelity. These data suggest that the antitermination complex and precursor RNA elements help to ensure quality control during ribosome biogenesis.
ABSTRACT

Ribosomes of Bacteroidia (formerly Bacteroidetes) fail to recognize Shine-Dalgarno (SD) sequences even though they harbor the anti-SD (ASD) of 16S rRNA. Inhibition of SD-ASD pairing is due to sequestration of the 3’ tail of 16S rRNA in a pocket formed by bS21, bS18, and bS6 on the 30S platform. Interestingly, in many Flavobacteriales, the gene encoding bS21, rpsU, contains an extended SD sequence. In this work, we present genetic and biochemical evidence that bS21 synthesis in Flavobacterium johnsoniae is autoregulated via a subpopulation of ribosomes that specifically lack bS21. Mutation or depletion of bS21 in the cell increases translation of reporters with strong SD sequences, such as rpsU’-gfp, but has no effect on other reporters. Purified ribosomes lacking bS21 (or its C-terminal region) exhibit higher rates of initiation on rpsUmRNA and lower rates of initiation on other (SD-less) mRNAs than control ribosomes. The mechanism of autoregulation depends on extensive pairing between mRNA and 16S rRNA, and exceptionally strong SD sequences, with predicted pairing free energies of < –13 kcal/mol, are characteristic of rpsU across the Bacteroidota. This work uncovers a clear example of specialized ribosomes in bacteria.
The Fredrick Lab made the trip out to Bordeaux, France to attend the Ribosome meeting (@2022Ribosomes). Kurt gave a talk, while Sepideh, Siddik, and Ben gave poster presentations. We were joined by our collaborator and fellow buckeye, Ralf Bundschuh. Everyone got great feedback and talked a lot of science! Not to mention there was a lot of good wine and food there too!






After 5 years of working in the Fredrick lab it’s time for this little birdy to fly! I have at long last learned the truth of the anti-Shine Dalgarno sequence…somewhat. Verily, I am grateful for all of the great things I have learned and experienced in the lab. Big thanks to Kurt and everyone else (Bappa, Michelle, Menglin, Lanqing, Ben, Sepideh, Fawwaz, Dean, Siddik, and Stephen).

Zakkary A. McNutt, Mai D. Gandhi, Elan A. Shatoff, Bappaditya Roy, Aishwarya Devaraj, Ralf Bundschuh, and Kurt Fredrick
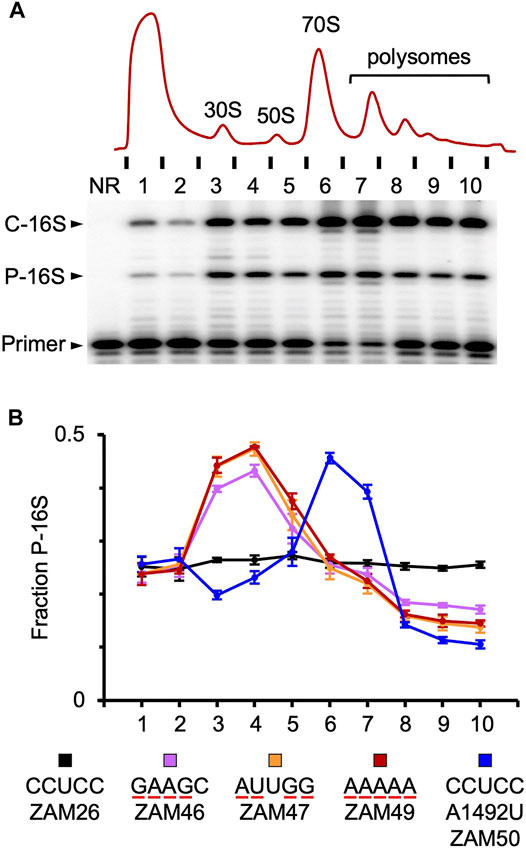
The anti-Shine-Dalgarno (ASD) sequence of 16S rRNA is highly conserved across Bacteria, and yet usage of Shine-Dalgarno (SD) sequences in mRNA varies dramatically, depending on the lineage. Here, we compared the effects of ASD mutagenesis in Escherichia coli, a Gammaproteobacteria which commonly employs SD sequences, and Flavobacterium johnsoniae, a Bacteroidia which rarely does. In E. coli, 30S subunits carrying any single substitution at positions 1,535–1,539 confer dominant negative phenotypes, whereas subunits with mutations at positions 1,540–1,542 are sufficient to support cell growth. These data suggest that CCUCC (1,535–1,539) represents the functional core of the element in E. coli. In F. johnsoniae, deletion of three ribosomal RNA (rrn) operons slowed growth substantially, a phenotype largely rescued by a plasmid-borne copy of the rrn operon. Using this complementation system, we found that subunits with single mutations at positions 1,535–1,537 are as active as control subunits, in sharp contrast to the E. coli results. Moreover, subunits with quadruple substitution or complete replacement of the ASD retain substantial, albeit reduced, activity. Sedimentation analysis revealed that these mutant subunits are overrepresented in the subunit fractions and underrepresented in polysome fractions, suggesting some defect in 30S biogenesis and/or translation initiation. Nonetheless, our collective data indicate that the ASD plays a much smaller role in F. johnsoniae than in E. coli, consistent with SD usage in the two organisms.
 Bappa, Zakk, and Kurt publish a paper in NAR titled, Structural basis of sequestration of the anti-Shine-Dalgarno sequence in the Bacteroidetes ribosome (2021). With collaborators from the Ortega lab at McGill University, Montreal, Quebec we solved the structure of the ribosome from Flavobacterium johnsoniae. Though SD sequences are heavily underrepresented among the Bacteroidetes, with the help of our collaborators at OSU, Elan Shatoff and Ralf Bundschuh, we show bioinformatically that the genes coding for 30S proteins S21 (rpsU) and S18 (rpsR) in many Bacteroidetes have perfect SD sequences upstream of their start codons, suggesting a novel mechanism of autoregulation.
Bappa, Zakk, and Kurt publish a paper in NAR titled, Structural basis of sequestration of the anti-Shine-Dalgarno sequence in the Bacteroidetes ribosome (2021). With collaborators from the Ortega lab at McGill University, Montreal, Quebec we solved the structure of the ribosome from Flavobacterium johnsoniae. Though SD sequences are heavily underrepresented among the Bacteroidetes, with the help of our collaborators at OSU, Elan Shatoff and Ralf Bundschuh, we show bioinformatically that the genes coding for 30S proteins S21 (rpsU) and S18 (rpsR) in many Bacteroidetes have perfect SD sequences upstream of their start codons, suggesting a novel mechanism of autoregulation.

Michelle, Ben, Menglin, and Kurt published a paper in JMB titled, Functional Analysis of BipA in E. coli Reveals the Natural Plasticity of 50S Subunit Assembly (2020).


Menglin and Kurt published a review article in the Journal of Molecular Biology titled, RNA Polymerase’s Relationship with the Ribosome: Not So Physical, Most of the Time (2020).
RNA Polymerase’s Relationship with the Ribosome: Not So Physical, Most of the Time – ScienceDirect

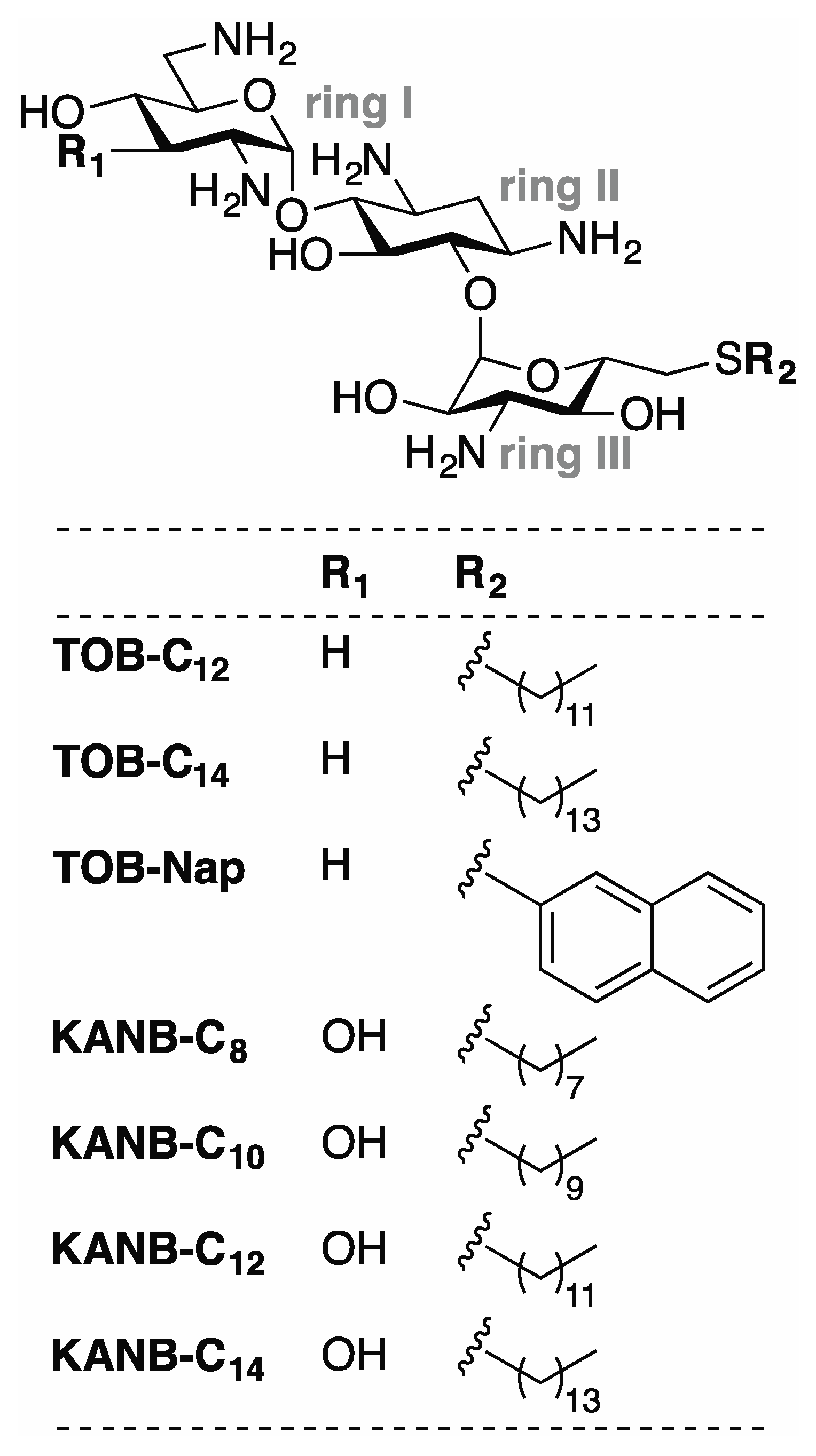 Lanqing, Hongkun, and Kurt published a paper in the journal of Antibiotics titled, Modified Aminoglycosides Bind Nucleic Acids in High-Molecular-Weight Complexes (2020).
Lanqing, Hongkun, and Kurt published a paper in the journal of Antibiotics titled, Modified Aminoglycosides Bind Nucleic Acids in High-Molecular-Weight Complexes (2020).
Modified Aminoglycosides Bind Nucleic Acids in High-Molecular-Weight Complexes – PubMed (nih.gov)