I’ve written a number of times on the skepticism and lack of understanding surrounding operational excellence (Op-ex) deployments in healthcare. Under the op-ex umbrella, there are several change management initiatives such as lean, Six Sigma, Total Quality Management, etc. All of these emphasize the importance of a systemic approach toward the sustainable improvement of key performance metrics.
Although there are several books and research on the benefits from these initiatives in healthcare context, skeptics dismiss them as mostly anecdotal and devoid of the rigor necessary for causal inferences (i.e., the presence of op-ex leads to healthcare improvements). In this regard, the skeptics are right. Many existing inferences in this area come from surveys (i.e., asking healthcare leaders about op-ex and correlating results to performance) or are based on secondary data that may not adequately capture op-ex elements.
Recent research Peter Ward & I conducted with Dr. Susan Moffatt-Bruce and other collaborators at The Ohio State University Wexner Medical Center leaves little room for skepticism about the impact of op-ex deployment in healthcare. Our quasi-experimental investigation permitted us to compare care delivery performance before and after deployment, using a control group from the same setting to account for any variations in the system beyond our experiment. Results show that op-ex deployment reduced 30-day readmissions, decreased monthly operating costs, improved patient satisfaction and also improved the quality of work among caregivers. In short, it works.
The Quasi-Experiment
Our experimental intervention was conducted over a period of three years and used op-ex principles to change the way patients were discharged after a kidney transplant. We specifically looked at a fundamental op-ex element: Standard work during patient discharge. This represents the set of activities standardized in terms of content, sequence, timing and outcome but still able to “flex” based on patient characteristics. An important feature of standard work is that it should be designed and developed by people doing the work and continuously monitored for improvement. In our study, more than 40 nurses, physicians, social workers, IT specialists and nurse managers responsible for discharging the patients designed and developed the new standard work over a nine-month period.
We focused on the kidney transplant discharge process because these recipients require prolonged periods of care after discharge. In fact, one in three U.S. kidney transplant patients is readmitted within 30 days, resulting in higher hospitalization costs, resource utilization (i.e. longer wait times for other wait-listed recipients), and – more importantly – poor quality of life for the patient post-procedure. When looking closely at the discharge process, we found patients were taking in an overwhelming amount of information in the four days prior to discharge. In this short period, caregivers covered more than 45 topics, including medication, infection prevention, lab compliance, fluid intake, exercise, and dental hygiene, among many others. Some of these instructions – dental hygiene, for one – were not critical to patients’ immediate well-being and could have been given post-discharge. Nurses, we found, also varied their delivery of instructions, adding to confusion and anxiety. Figure 1 represents the current state of discharge process prior to our intervention. As seen from this figure, variability during the discharge process confused the patients and heightened anxiety levels after discharge, triggering readmissions.
The new standardized discharge work sought to resolve these inconsistencies. The caregiving team developed specific instructions on the content, sequence, timing, and delivery outcome for each element of discharge, drawing from data on 15 former transplant patients. The team also decided to adopt a two-part instructional approach, with inpatient nurses giving the most essential discharge instructions during the hospital stay (Part I Instructions), while outpatient nurses gave other instructions (Part II Instructions) 48 hours after discharge. This resulted in a formal handoff process between the inpatient and outpatient caregiving teams. After implementing this new process, the team also adopted 10-minute “huddles,” or standing meetings, to continuously monitor and improve communication at inpatient (bi-weekly) and outpatient (weekly) units by focusing on problem solving and best practice-sharing. These huddles have been going on for more than a year, with full participation from the caregiving teams. The entire op-ex implementation, launched in May 2015, took about a year. Figure 2 represents the process after the op-ex implementation.
To validate the benefits of the Op-Ex implementation, we tracked the discharge process outcomes for more than 700 kidney transplant patients for four years (two years prior to, and one year after, implementation). To ensure our data weren’t skewed by factors such as discharge procedure changes or electronic medical record implementation, we collected patient discharge process data from 180 heart and liver transplants, using them as a control group. Patients undergoing heart and liver transplants experience similar outpatient issues as kidney transplant recipients and have very similar discharge instructions (as seen in Figure 1). We also controlled for patient factors such as age, preexisting conditions, length of stay, functioning of transplanted organ at the time of discharge, ethnicity, 30-day mortality rates, etc. In addition, we adjusted for any other changes to the process that were not a part of our experiment (e.g. adding new technologies)
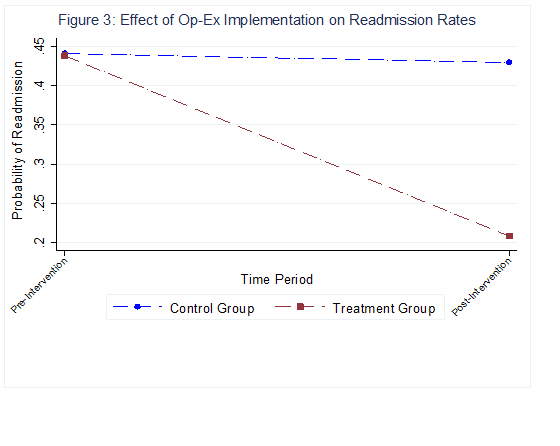
After accounting for all these effects, we used a difference-in-difference (DID) approach to estimate the causal effects of our implementation on readmission outcomes. Our analyses suggest that the likelihood of getting readmitted to the hospital was about 35% lower for the treatment group (kidney transplant process after Op-Ex implementation) compared to the control group and pre-implementation group (See Figure 3). On average, this accounted to a 25% reduction in the overall readmission rates after the intervention for the treatment group, resulting in a conservative savings estimate of $85,000 due to hospitalization expenses for the unit. This doesn’t capture savings from the overall patient well- being after the surgery.
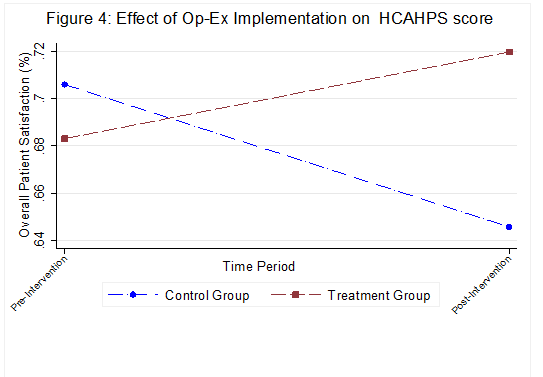
We also found a 10% increase in the overall patient experience quality score, measured using the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey. Interestingly, the control group unit experienced an 11% decline in these scores during the same time period (see Figure 4). Improving the scores from this survey has been a key objective for hospital administrators as Medicare/Medicaid reimbursements are now tied to them.
Our study shows that Op-Ex transformation indeed can result in improvement across multiple outcomes. The real challenge is how to initiate and sustain this journey. All these results were possible due to the hard work of all the front-line nurses in this unit. They truly are the miracle workers who continue to sustain this journey and innovate as they move along.